SNO WORKSHOP I on NANOCERIA
November 2, 2013
WORKSHOP SPONSORED by University of Kentucky and SNO

| Tracy Farmer Institute for Sustainability
and the Environment |
Associate Dean for Research |
| Department of Pharmaceutical Sciences | Office of the Vice President for Research |
| College of Pharmacy | |
 Panel 1 topic: The current and proposed uses of nanoceria, and the basis for their use (properties) such as its catalytic activity as a fuel additive and in fuel cells; insoluble abrasive for mechanical planarization; and redox for anti-oxidative, anti-inflammatory therapeutics. Where is it being used? Why do people care?
Panel 1 topic: The current and proposed uses of nanoceria, and the basis for their use (properties) such as its catalytic activity as a fuel additive and in fuel cells; insoluble abrasive for mechanical planarization; and redox for anti-oxidative, anti-inflammatory therapeutics. Where is it being used? Why do people care?
Summary: Cerium dioxide is an extremely versatile and powerful catalytic material, owing to its intrinsic ability to form oxygen vacancies and facilitate redox chemistry. This pliable material can form a variety of simple or complex structures and its properties can be further tailored by substitutional metal doping, thus making it useful in a plethora of commercial areas, including chemical polishing and planarization, fuel cells, automotive catalytic converters, and even therapeutic applications.
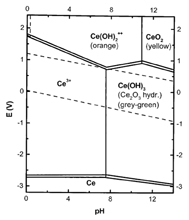 Panel 2 topic: Does nanoceria behave both as a pro-and anti-oxidant depending on its synthesis, physico-chemical properties, dispersion, and environmental conditions?
Panel 2 topic: Does nanoceria behave both as a pro-and anti-oxidant depending on its synthesis, physico-chemical properties, dispersion, and environmental conditions?
Summary: The redox properties of nanoceria depend on particle size, the concentration of vacancies near the surface, the nanoparticle surface chemistry (including surfactants, salts, coupling agents, proteins, and other ligands), the pH of the environment, and ions or ligands available in its aqueous environment.
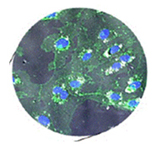 Panel 3 topic: What are the salient physico-chemical properties (size, surface chemistry, stabilizer) contributing to the potential positive and negative aspects of nanoceria in a biological system?
Panel 3 topic: What are the salient physico-chemical properties (size, surface chemistry, stabilizer) contributing to the potential positive and negative aspects of nanoceria in a biological system?
Summary: Nanoceria at the biological interface. Picture showing ultra fine ceria nanoparticles modified by biological cells. Redox chemistry of nanoceria changes as a function of its surrounding environment – showing the dynamic nature of the nanoparticle in a complex biological environment.
 Panel 4 topic: The biological identity of nanoceria and its influence on the biological response.
Panel 4 topic: The biological identity of nanoceria and its influence on the biological response.
Summary: Nanoceria that enter the body are immediately coated with biomolecules that influence the subsequent biological response. Future studies should take the biological environment into account in order to understand and manipulate the biological response to nanoceria.
 Panel 5 topic: The Yang: Potential therapeutic development of nanoceria.
Panel 5 topic: The Yang: Potential therapeutic development of nanoceria.
Summary:
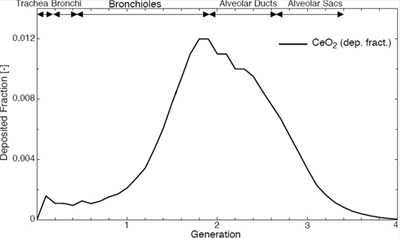 Panel 6 topic: The Yin: The toxicological risks of nanoceria: accumulation, target organs and issues of clearance.
Panel 6 topic: The Yin: The toxicological risks of nanoceria: accumulation, target organs and issues of clearance.
Summary: Nanoceria persists at the site of its accumulation or injection. Lung uptake is low and gastrointestinal uptake is extremely low. Adverse effects have been characterized with high exposures. The boundary between no and low effect has not been well established.
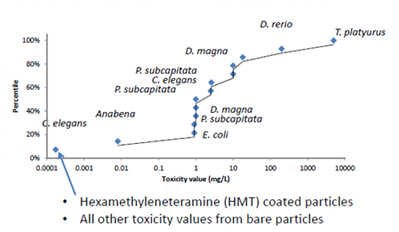 Panel 7 topic: Life cycle analysis, and environmental fate and transport of ceria.
Panel 7 topic: Life cycle analysis, and environmental fate and transport of ceria.
Summary:
• Risk to aquatic and terrestrial receptors from diesel fuel additive likely negligible
• Little information available on exposures from other uses
• Some aquatic receptor species sensitive at low ug/L concentrations
• Hexamethyleneteramine coated particles far more toxic than other particle types